A human iPSC cerebral organoid in which pigmented retinal epithelial cells can be seen (from the work of McClure-Begley et al.). Also see “Can lab-grown brains become conscious?” by Sara Readon Nature 2020.
The fact that experiments on people are severely constrained is a major obstacle in understanding human development and disease. Some of these constraints are moral and ethical and clearly appropriate and necessary given the depressing history of medical atrocities. Others are technical, associated with the slow pace of human development. The combination of moral and technical factors has driven experimental biologists to explore the behavior of a wide range of “model systems” from bacteria, yeasts, fruit flies, and worms to fish, frogs, birds, rodents, and primates. Justified by the deep evolutionary continuity between these organisms (after all, all organisms appear to be descended from a single common ancestor and share many molecular features), experimental evolution-based studies of model systems have led to many therapeutically valuable insights in humans – something that I suspect a devotee of intelligent design creationism would be hard pressed to predict or explain (post link).
While humans are closely related to other mammals, it is immediately obvious that there are important differences – after all people are instantly recognizable from members of other closely related species and certainly look and behave differently from mice. For example, the surface layer of our brains is extensively folded (they are known as gyrencephalic) while the brain of a mouse is smooth as a baby’s bottom (and referred to as lissencephalic). In humans, the failure of the brain cortex to fold is known as lissencephaly, a disorder associated with severe neurological defects. With the advent of more and more genomic sequence data, we can identify human specific molecular (genomic) differences. Many of these sequence differences occur in regions of our DNA that regulate when and where specific genes are expressed. Sholtis & Noonan (1) provide an example: the HACNS1 locus is a 81 basepair region that is highly conserved in various vertebrates from birds to chimpanzees; there are 13 human specific changes in this sequence that appear to alter its activity, leading to human-specific changes in the expression of nearby genes (↓). At this point ~1000 genetic elements that are different in humans compared to other vertebrates have been identified and more are likely to emerge (2). Such human-specific changes can make modeling human-specific behaviors, at the cellular, tissue, organ, and organism level, in non-human model systems difficult and problematic (3, 4). It is for this reason that scientists have attempted to generate better human specific systems.

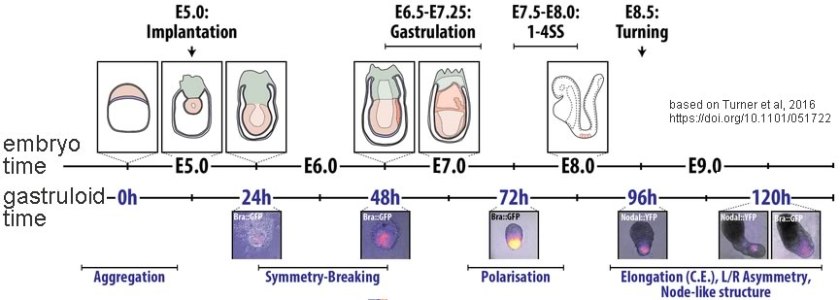
One particularly promising approach is based on what are known as embryonic stem cells (ESCs) or pluripotent stem cells (PSCs). Human embryonic stem cells are generated from the inner cell mass of a human embryo and so involve the destruction of that embryo – which raises a number of ethical and religious concerns as to when “life begins” (5). Human pluripotent stem cells are isolated from adult tissues but in most cases require invasive harvesting methods that limit their usefulness. Both ESCs and PSCs can be grown in the laboratory and can be induced to differentiate into what are known as gastruloids. Such gastruloids can develop anterior-posterior (head-tail), dorsal-ventral (back-belly), and left-right axes analogous to those found in embryos (6) and adults (top panel ↓). In the case of PSCs, the gastruloid (bottom panel ↓) is essentially a twin of the organism from which the PSCs were derived, a situation that raises difficult questions: is it a distinct individual, is it the property of the donor or the creation of a technician. The situation will be further complicated if (or rather, when) it becomes possible to generate viable embryos from such gastruloids.

 The Nobel prize winning work of Kazutoshi Takahashi and Shinya Yamanaka (7), who devised methods to take differentiated (somatic) human cells and reprogram them into ESC/PSC-like cells, cells known as induced pluripotent stem cells (iPSCs)(8), represented a technical breakthrough that jump-started this field. While the original methods derived sample cells from tissue biopsies, it is possible to reprogram kidney epithelial cells recovered from urine, a non-invasive approach (9, 10). Subsequently, Madeline Lancaster, Jurgen Knōblich, and colleagues devised an approach by which such cells could be induced to form what they termed “cerebral organoids” (although Yoshiki Sasai and colleagues were the first to generate neuronal organoids); they used this method to examine the developmental defects associated with microencephaly (11). The value of the approach was rapidly recognized and a number of studies on human conditions, including lissencephaly (12), Zika-virus infection-induced microencephaly (13), and Down’s syndrome (14); investigators have begun to exploit these methods to study a range of human diseases – and rapid technological progress is being made.
The Nobel prize winning work of Kazutoshi Takahashi and Shinya Yamanaka (7), who devised methods to take differentiated (somatic) human cells and reprogram them into ESC/PSC-like cells, cells known as induced pluripotent stem cells (iPSCs)(8), represented a technical breakthrough that jump-started this field. While the original methods derived sample cells from tissue biopsies, it is possible to reprogram kidney epithelial cells recovered from urine, a non-invasive approach (9, 10). Subsequently, Madeline Lancaster, Jurgen Knōblich, and colleagues devised an approach by which such cells could be induced to form what they termed “cerebral organoids” (although Yoshiki Sasai and colleagues were the first to generate neuronal organoids); they used this method to examine the developmental defects associated with microencephaly (11). The value of the approach was rapidly recognized and a number of studies on human conditions, including lissencephaly (12), Zika-virus infection-induced microencephaly (13), and Down’s syndrome (14); investigators have begun to exploit these methods to study a range of human diseases – and rapid technological progress is being made.
The production of cerebral organoids from reprogrammed human somatic cells has also attracted the attention of the media (15). While “mini-brain” is certainly a catchier name, it is a less accurate description of a cerebral organoid, itself possibly a bit of an overstatement, since it is not clear exactly how “cerebral” such organoids are. For example, the developing brain is patterned by embryonic signals that establish its asymmetries; it forms at the anterior end of the neural tube (the nascent central nervous system and spinal cord) and with distinctive anterior-posterior, dorsal-ventral, and left-right asymmetries, something that simple cerebral organoids do not display. Moreover, current methods for generating cerebral organoids involve primarily what are known as neuroectodermal cells – our nervous system (and that of other vertebrates) is a specialized form of the embryo’s surface layer that gets internalized during development. In the embryo, the developing neuroectoderm interacts with cells of the circulatory system (capillaries, veins, and arteries), formed by endothelial cells and what are known as pericytes that surround them. These cells, together with interactions with glial cells (astrocytes, a non-neuronal cell type) combine to form the blood brain barrier. Other glial cells (oligodendrocytes) are also present; in contrast, both types of glia (astrocytes and oligodendrocytes) are rare in the current generation of cerebral organoids. Finally, there are microglial cells, immune system cells that originate from outside the neuroectoderm; they invade and interact with neurons and glia as part of the brain’s dynamic neural  system. The left panel of the figure shows, in highly schematic form how these cells interact (16). The right panel is a drawing of neural tissue stained by the Golgi method (17), which reveals ~3-5% of the neurons present. There are at least as many glial cells present, as well as microglia, none of which are visible in the image. At this point, cerebral organoids typically contain few astrocytes and oligodendrocytes, no vasculature, and no microglia. Moreover, they grow to be about 1 to 3 mm in diameter over the course of 6 to 9 months; that is significantly smaller in volume than a fetal or newborn’s brain. While cerebral organoids can generate structures characteristic of retinal pigment epithelia (top figure) and photo-responsive neurons (18), such as those associated with the retina, an extension of the brain, it is not at all clear that there is any significant sensory input into the neuronal networks that are formed within a cerebral organoid, or any significant outputs, at least compared to the role that the human brain plays in controlling bodily and mental functions.
system. The left panel of the figure shows, in highly schematic form how these cells interact (16). The right panel is a drawing of neural tissue stained by the Golgi method (17), which reveals ~3-5% of the neurons present. There are at least as many glial cells present, as well as microglia, none of which are visible in the image. At this point, cerebral organoids typically contain few astrocytes and oligodendrocytes, no vasculature, and no microglia. Moreover, they grow to be about 1 to 3 mm in diameter over the course of 6 to 9 months; that is significantly smaller in volume than a fetal or newborn’s brain. While cerebral organoids can generate structures characteristic of retinal pigment epithelia (top figure) and photo-responsive neurons (18), such as those associated with the retina, an extension of the brain, it is not at all clear that there is any significant sensory input into the neuronal networks that are formed within a cerebral organoid, or any significant outputs, at least compared to the role that the human brain plays in controlling bodily and mental functions.
The reasonable question, then, must be whether a cerebral organoid, which is a relatively simple system of cells (although itself complex), is conscious. It becomes more reasonable as increasingly complex systems are developed, and such work is proceeding apace. Already researchers are manipulating the developing organoid’s environment to facilitate axis formation, and one can anticipate the introduction of vasculature. Indeed, the generation of microglia-like cells from iPSCs has been reported; such cells can be incorporated into cerebral organoids where they appear to respond to neuronal damage in much the same way as microglia behave in intact neural tissue (19).
We can ask ourselves, what would convince us that a cerebral organoid, living within a laboratory incubator, was conscious? How would such consciousness manifest itself? Through some specific pattern of neural activity, perhaps? As a biologist, albeit one primarily interested in molecular and cellular systems, I discount the idea, proposed by some physicists and philosophers as well as the more mystical, that consciousness is a universal property of matter (20,21). I take consciousness to be an emergent property of complex neural systems, generated by evolutionary mechanisms, built during embryonic and subsequent development, and influenced by social interactions (BLOG LINK) using information encoded within the human genome (something similar to this: A New Theory Explains How Consciousness Evolved). While a future concern, in a world full of more immediate and pressing issues, it will be interesting to listen to the academic, social, and political debate on what to do with mini-brains as they grow in complexity and perhaps inevitably, towards consciousness.
Footnotes and references
Thanks to Rebecca Klymkowsky, Esq. and Joshua Sanes, Ph.D. for editing and disciplinary support. Minor updates and the reintroduction of figures 22 Oct. 2020.
- Gene regulation and the origins of human biological uniqueness
- See also Human-specific loss of regulatory DNA and the evolution of human-specific traits
- The mouse trap
- Mice Fall Short as Test Subjects for Some of Humans’ Deadly Ill
- The status of the human embryo in various religions
- Interactions between Nodal and Wnt signalling Drive Robust Symmetry Breaking and Axial Organisation in Gastruloids (Embryonic Organoids)
- Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors
- How iPS cells changed the world
- Generation of Induced Pluripotent Stem Cells from Urine
- Urine-derived induced pluripotent stem cells as a modeling tool to study rare human diseases
- Cerebral organoids model human brain development and microcephaly.
- Human iPSC-Derived Cerebral Organoids Model Cellular Features of Lissencephaly and Reveal Prolonged Mitosis of Outer Radial Glia
- Using brain organoids to understand Zika virus-induced microcephaly
- Probing Down Syndrome with Mini Brains
- As an example, see The Beauty of “Mini Brains”
- Derived from Central nervous system pericytes in health and disease
- Golgi’s method .
- Cell diversity and network dynamics in photosensitive human brain organoids
- Efficient derivation of microglia-like cells from human pluripotent stem cells
- The strange link between the human mind and quantum physics – BBC:
- Can Quantum Physics Explain Consciousness?

“I take consciousness to be an emergent property of complex neural systems, generated by evolutionary mechanisms, built during embryonic and subsequent development, and influenced by social interactions (BLOG LINK) using information encoded within the human genome (something similar to this: A New Theory Explains How Consciousness Evolved). ” This is just a fancy way of saying I don’t know the origins of consciousness. Have some courage. Admit you do not know. Emergent??? Indeed.
LikeLike
A great model! It should be particularly valuable for testing early development by optogenetic methods.
I hope you don’t mind that I have translated your article in German and put it online on my blog:
http://scienceblog.at/soll-man-sich-sorgen-machen-dass-menschliche-mini-hirne-bewusstsein-erlangen#.
LikeLike